What are the effects of light pollution on insect communities on the Davidson College campus?
To answer this question, we selected six sites on campus, three in continuously lit areas, usually near parking lots, and three sites that were unlit. All sites were in grassy areas near the edge of a patch of woods. We used light traps, which were white sheets strung tightly in a PVC pipe frame, in front of which we hung a UV light attached to a battery (see image to the right). Each sheet was subdivided into 16 panels. At 2 a.m. on collection nights, the researchers (three undergraduate students and one professor) would inspect the trap and take images using a high resolution Canon DSLR EOS D60 camera. Images were taken of each panel, which were processed and analyzed back at the laboratory. Insects on the trap were inspected; unusual or unknown insects were collected and brought back to the laboratory, where they were frozen and later identified to the lowest taxonomic resolution. From those identifications, insects in the images were often able to be identified, although small insects, even if known from specimen inspection, were often unidentifiable in digital images.
These images to the right provide a sense of our data analysis process. We began with an image. Sometimes the images, or parts of the image, were out of focus, because either insects were caught in motion or the sheet was slightly wrinkled and not completely flat. In those cases, multiple images were used to zoom in on individual insects to identify them to the lowest taxonomic resolution.

Image of one panel of the light trap with several small insects on it, after it was processed, revealing the proposed identifications of the various insects.
For most images, if not too many insects were on it, students labeled individual specimens with the proposed identification. In that way, quality control of the data could be maintained, and the professor could revisit any identification to verify or correct. All in all, there were 5 trap nights at each of six sites, with 16 panel images for each trap night, leading to a minimum of 480 images reviewed (minimum because some panels had multiple copies reviewed for clarification.
Overall, we found that insect abundance is far greater in dark areas on campus than in continuously lit areas. Insect abundance increases as temperature increases, but increases far faster for sites in the dark than in lit areas. In the dark, insect abundance increases by 200 insects per degree increase in temperature, while in the light, abundance increases by only 41 insects per degree increase. Both of these slopes are significantly different from zero, as shown in the top portion of the table below, and they are significantly different from each other.

Insect abundance increases as temperature increases, but increases far faster for sites in the dark than in lit areas.

Table of regression results. For each test in the left column, there is a test of whether the slope for dark and light are different from a slope of zero, as well as a test of whether those two slopes are different from each other.
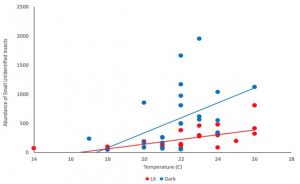
Figure of small unidentified insect abundance at different temperatures for dark and lit sites. Abundance increased faster in dark sites.
Regression results also revealed that the abundance of small unidentified insects, comprised mostly of parasitoid wasps (O. Hymenoptera), various families of small flies and midges (O. Diptera), and small hoppers (O. Hemiptera), followed the same trend as total insect abundance. Or rather, total abundance followed this trend, as small unidentified insects made up a large fraction of total insects. To understand that, compare the figure above with the one to the right.
Total moth abundance increased significantly with temperature only at the dark sites. At the lit sites, while the slope was slightly positive, statistically it was no different from a slope of zero. The slopes of dark and lit sites were significantly different from one another; again, see the table for the statistical results. While there is a lot of scatter in the raw data graph, keep in mind that the statistical tests were performed on the log-transformed abundances.
The trends in scarab beetle abundance were similar the trends in total moth abundance (see figure to the right). Abundances of another family of beetles, the rove beetles (F. Staphylinidae) were not affected by temperature. However, rove beetles were about 10x more abundant at dark site traps than at lit site traps, and this was consistent throughout the summer (see bar chart below).

Rove beetle abundance was not affected by night-time temperature but was greater at dark than at lit sites.
A few interesting species we’ve observed:
Return to Dr. Paradise’s home page









